The global pharmaceutical industry loses billions every year due to temperature excursions during storage and transit. A single temperature breach, even a few degrees above the specified range, can render a life-saving medicine, vaccine, biologic, or insulin completely ineffective. Undetected exposure to temperature of pharmaceutical products and their patient administration can have adverse consequences. This is why pharmaceutical cold chain monitoring has become one of the critical operational priorities for pharmaceutical manufacturers, distributors, hospitals, and pharmacies worldwide.
In this blog we discuss in detail what pharmaceutical cold chain monitoring really is, why is it important, why it works, what devices are used, common challenges in pharmaceutical cold chain monitoring and how Lisaline, the supplier of cold chain monitoring solutions in India, solves these challenges. We also discuss the role of wireless data loggers with cloud integration, IoT-based real-time monitoring systems and temperature indicators in pharmaceutical cold chain monitoring. At the end of this blog, you will know everything about pharmaceutical cold chain monitoring.
What is Pharmaceutical Cold Chain Monitoring?
Pharmaceutical cold chain monitoring is the continuous process of tracking, recording and managing environmental conditions like temperature and sometimes humidity throughout the entire supply chain of temperature-sensitive pharmaceutical products.
It begins the moment a drug or vaccine leaves the manufacturing facility and continues through warehousing, transportation, and final delivery to the end user. The end user might be the hospital, pharmacy or patient.
Pharmaceutical cold chain monitoring doesn’t just involve keeping the products within their suitable environmental conditions. The process involves real-time data logging, threshold-based real-time alerts, regulatory documentation, and audit-ready reporting. This is done to prove that products have remained within their specified temperature ranges at various stages of the supply chain.
Lisaline Asia is the leading supplier of cold chain monitoring solutions in India. We provide various devices for your smooth pharmaceutical cold chain monitoring. These include irreversible temperature indicators, LoRaWAN-enabled wireless data loggers, USB data loggers, 4G data loggers, shipment data loggers, Bluetooth data loggers, and temperature-controlled cold chain boxes. These solutions combined ensure that your pharmaceutical products stay within a suitable range and you get notified immediately in case of excursions. These ensure you get to know in case there is a breach, and affected products are not administered to facilities or patients.
Why is cold chain monitoring important for pharmaceuticals?
Pharmaceutical cold chain monitoring is not just an optional process. It is a regulatory, ethical and commercial necessity in modern pharma supply chains. Here are the reasons why cold chain monitoring is important for pharmaceutical products:
Patient Safety
The most important reason why pharmaceutical cold chain monitoring is important is patient safety. A critical medicine or vaccine may be exposed to temperatures above its safe thresholds. Their potency may have been affected, but they may appear intact physically. A patient who receives this vaccine or medicine may feel they are protected, but the product might be life-threatening. Therefore, it is important that the pharmaceutical suppliers get a clear signal if a product has been exposed to unsafe temperatures.
Regulatory Compliance
Global regulatory bodies mandate strict cold chain documentation and monitoring.
- WHO publishes guidelines on Good Distribution Practice (GDP) for pharmaceutical products.
- US FDA (21 CFR Part 11) requires electronic records and audit trails for drug storage.
- EU GDP Guidelines (2013/C 343/01) mandates continuous temperature monitoring and real-time alerts.
- India CDSCO increasingly aligning with international GDP standards for cold chain logistics.
- IATA and ICAO regulate temperature-sensitive air cargo (pharma is a dominant category)
Non compliance to these standards can lead to product recalls, loss of licenses, regulatory penalties and significantly reputational damage.
Financial Loss Prevention
Due to exposure to unsafe temperatures, the pharmaceutical industry loses billions. Many critical medicines, vaccines have to be discarded because their potency has been affected. This causes massive financial loss to pharma companies. Therefore, pharmaceutical cold chain monitoring is necessary. With modern technologies like cloud integration and real-time alerts, breaches can be prevented before they occur, minimising financial losses. Logistics teams can proactively monitor the shipments to ensure immediate actions are taken to prevent a breach.
Improving efficiency of the supply chain
Cold Chain Monitoring for medicines creates a verifiable chain of custody with timestamped data. This means that if an excursion occurs, it can be pinpointed, patterns can be clearly identified. Whether the breach occured at the warehouse, during transit, at a customs checkpoint, or at the last-mile delivery stage. This helps pharmaceutical companies to make improvements, reroute shipments and avoid excursions and losses in the future.
What are the different devices used in pharmaceutical cold chain monitoring?
Pharmaceutical cold chain monitoring includes a range of devices. These are designed for temperature and environmental monitoring at every stage of the supply chain from manufacturing to patient administration. From giving real time alerts to giving a clear visual indication of temperature exposure, these devices help in proactive cold chain management. These are designed to suit the different requirements of pharmaceutical facilities.
Temperature Indicators
Lisaline Asia supplies single-use temperature indicators from Zebra Technologies. These are self-adhesive labels applied to the medicine bottles that monitor the temperature of the product continuously once they are applied. Temperature indicator labels include HEATmarker, Safe-T-Vue, LIMITmarker, FREEZEmarker. These provide a clear irreversible visual indication when the product is exposed to temperatures not matching the safe thresholds. These give a proof of temperature exposure and prevent the administration of unsafe products to patients.
LoRaWAN wireless data loggers
Lisaline Asia supplies wireless data loggers from Dickson. These include the Cobalt X Series Data Loggers. These multi-use data loggers support multiple sensors and measure temperature continuously throughout the supply chain at regular intervals. These also measure several other parameters, including humidity. Then the readings are sent through LoRaWAN connectivity to the OCEAView cloud platform.
Here, the readings can be viewed in real time either from the app or a web-based platform from a mobile device. This ensures that the logistics teams can spot breaches early and act on them, preventing loss. The cloud platform also provides real-time alerts on mobile through SMS, email or app-based notifications, whichever is selected, which ensures proactive actions reducing excursions. Moreover, the OCEAView platform allows easy downloading of time-stamped reports and exporting data, which helps in compliance and regulation in further decision-making.
LoRaWAN networks support long-range data transmission, hence you can monitor the parameters practically even if you are not with the shipment or in the warehouse where the products are stored. The alerts can also be received and acknowledged from the data logger device also.

USB Data Loggers
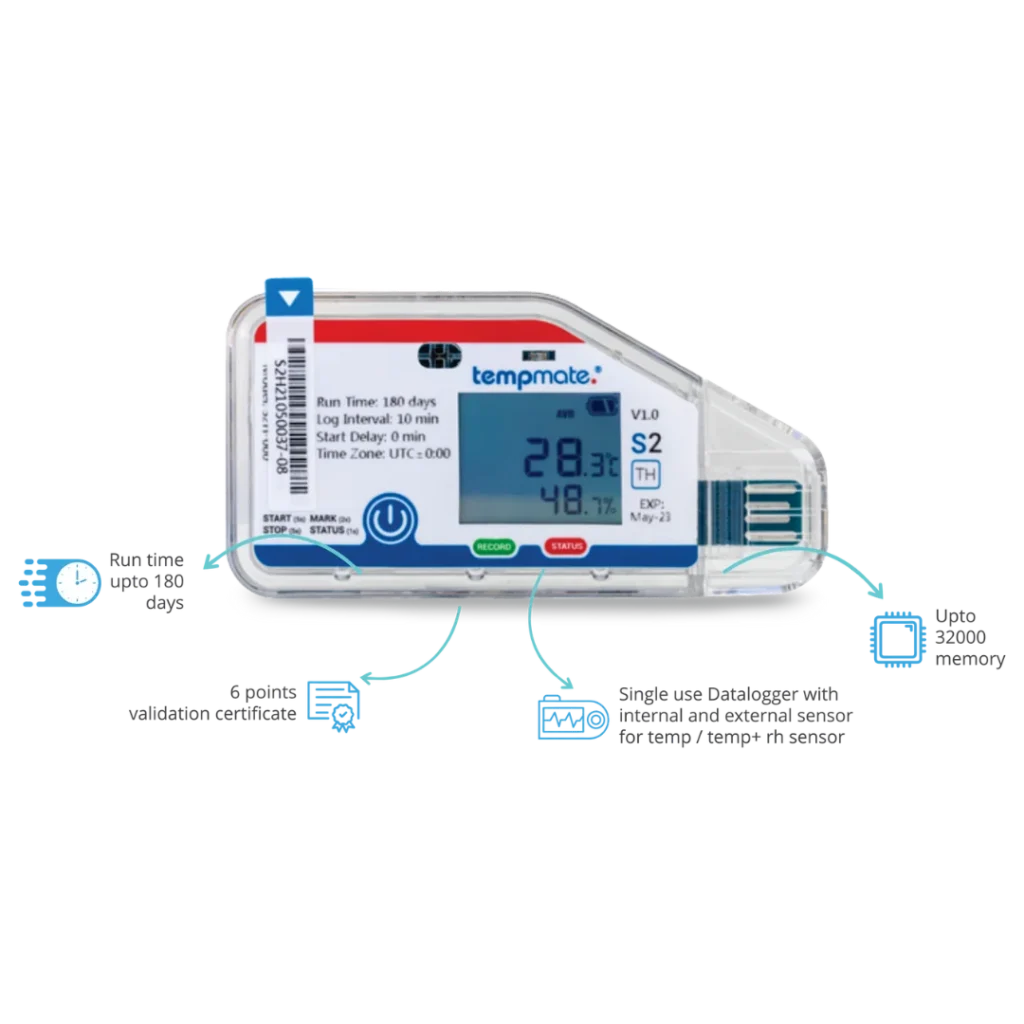
Lisaline supplies a USB data logger from Degrite. The Degrite S USB data logger is a single use disposable data logger. It monitors temperature with a single sensor throughout the supply chain journey. The PDF data logger features a plug and play display. This means that once the monitoring is done, you can plug the data logger to a USB port on a computer device. This will automatically generate time-stamped PDF/CSV reports and allow you to export data.

GSM Data Loggers
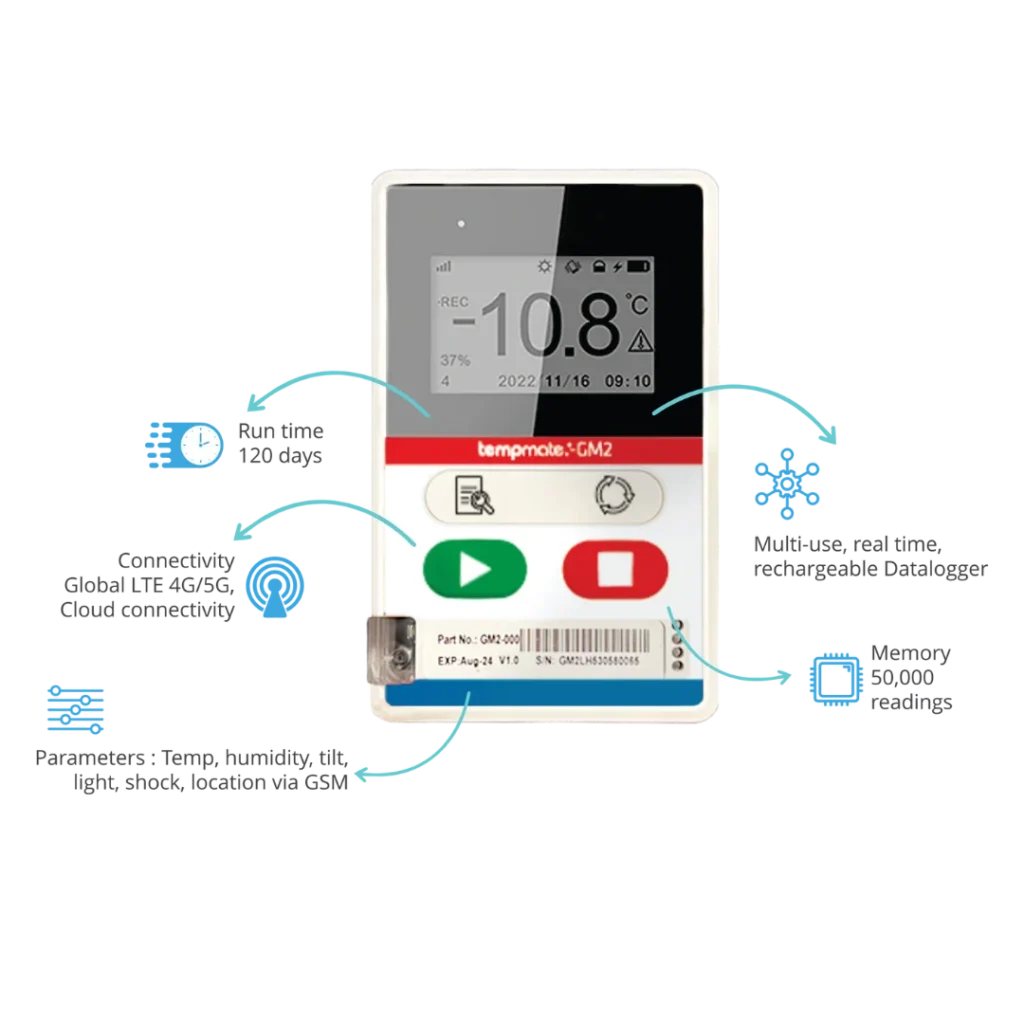
GSM Data Loggers are advanced 4G data loggers. These data loggers from Tempmate—Tempmate GS2 and Tempmate GM2 are single-use and multi-use data loggers that measure multiple parameters like temperature, humidity, light, shock, location, tilt, etc.
GSM data loggers use GSM networks (Global System for Mobile Connectivity). They transmit data from 4G connectivity with 2G fallback to the Tempmate cloud platform in real time. Here you can view live data, readings, graphs, download PDF/CSV reports, and get real-time alerts- SMS, email or others, whichever is selected on your mobile device. This ensures proactive cold chain management.
Global connectivity means that you can view the data literally from anywhere across the world, even if the shipment or warehouse is in a different country. If logistics teams take proactive and immediate actions on the alerts sent, which are locally indicated by the data logger device, also, then many excursions can be prevented.
Bluetooth Data Loggers
Lisaline Asia supplies Bluetooth-enabled data logging devices from Dickson and Zebra. Bluetooth data loggers are wireless temperature sensors that can function independently as data loggers, too. The Bluetooth data loggers include Emerald and Atlas data logger models from Dickson, and the ZS300 model from Zebra. These transmit the readings from Bluetooth to cloud platforms. However, they can be viewed only in the Bluetooth range of the data logger device. Also the device should be connected by Bluetooth to the data logger device. From the cloud platform, you can view real time data, download PDF/CSV reports and get alerts in case of excursions.
Shipment Data Loggers with Display
Lisaline Asia supplies data loggers designed specifically for shipments with LCD displays. The LCD display shows the current readings like temperature, humidity, battery level and other essential information that would be helpful for logistics teams to track during shipment. These display data loggers have a built-in USB interface, which means that post shipment, these data loggers can be plugged into any device via USB, and PDF/CSV reports can be downloaded automatically along with data exporting.
Cold Chain Boxes
Lisaline supplies cold chain boxes from Hemokool. These cooling boxes for shipping operate on active and passive cooling systems. The Hemokool Active Cooling Boxes use the active cooling system for temperature control. These have precise electric controls and work as portable refrigerators and freezers for controlling the temperature of sensitive goods during transport. The Hemokool passive cooling boxes use a passive cooling system with non-electric operation. They rely on insulated design and pre-frozen coolant packs to keep the products cool for short-term shipments and temporary storage. Both these cold chain boxes ensure the temperature of the medicines and vaccines they carry remains safe throughout the supply chain.

What are the common challenges faced in pharmaceutical cold chain monitoring?
Pharmaceutical cold chain monitoring is essential for maintaining the quality and safety of temperature-sensitive products throughout storage and transport. However, this process comes with several challenges that must be managed carefully to prevent temperature excursions, product damage, and compliance issues:
Last-minute delivery
The infrastructure gap in last-minute delivery is one of the major challenges in pharmaceutical cold chain monitoring. The final leg of delivery from a regional distribution hub to a hospital, clinic, pharmacy, or patient’s home is where cold chain integrity most frequently breaks down. This occurs because last-mile failures still occur due to inadequate refrigerated transport, unreliable courier practices, and a lack of temperature monitoring on delivery vehicles. The problem is compounded by poor road infrastructure, frequent power outages, limited access to refrigeration equipment, and a shortage of trained cold chain personnel. Eg- A vaccine that stayed between 2°C and 8°C for weeks can be ruined in a 45-minute bike ride in summer heat.
Power gaps
Most cold chain storage equipment needs electricity to keep operating. Even a power outage for a few minutes, the temperature of the cold chain facility can rise rapidly. Many temperature monitoring systems also use electricity, so you can’t detect if the temperature has risen. The alarm systems also shut down during power outages. Nobody knows medicines are warming up. By the time staff arrive in the morning, the damage may already be done, and the medicines may be unusable and still appear normal physically.
Information trapped in different monitoring systems
Throughout the supply chain, different monitoring systems may be used at each stage of the product journey. This can make it difficult to get a complete picture of the temperature data and determine whether the product remained safe throughout transit and storage. If one system is inefficient or not properly calibrated, a temperature excursion may go undetected.
From manufacturer to supplier, supplier to retailer, and retailer to patient, multiple monitoring methods may be involved. As a result, it becomes challenging to pinpoint where an excursion occurred or identify recurring patterns that could help improve the process, such as rerouting shipments or adjusting handling procedures.
Manual Logs
Many cold chain facilities still rely on staff members physically walking up to a thermometer, reading the number, and writing it on a paper chart a few times a day. This creates huge blind spots. Eg- If the temperature spiked at 2 a.m. and came back down by 6 a.m., the paper log will show normal readings at both check-in times, and the problem will be completely invisible. Paper logs are also easy to make mistakes on or even to fill in without actually checking.
Inaccurate Sensors
Many inefficient pharmaceutical cold chain monitoring systems have inaccurate sensors. The sensors may show huge differences in temperature and other parameters, rendering the readings inaccurate and ineffective. Everything looks fine, but the medicine is being stored outside safe limits. This is why sensors need to be sent for calibration regularly (usually once a year) to check they are still accurate. Many organisations skip or delay calibration because it costs money and takes the sensor out of service for a while.
System Validation is Expensive and Slow
In most industries, you buy a piece of equipment, plug it in, and start using it. In the pharmaceutical industry, that is simply not allowed. Before any monitoring system can be used to generate compliance data, you must formally prove with documented evidence that it works correctly, consistently, and reliably.
This structured process, known as validation, happens in three stages. First, Installation Qualification (IQ) checks that everything is set up correctly. Then Operational Qualification (OQ) tests that the system does exactly what it claims, alerts on time, battery backup kicks in, and cloud data matches sensor readings. Finally, Performance Qualification (PQ) proves the system keeps working reliably under real-world conditions over several weeks.
Each stage demands pre-written test protocols, live documented results, and formal sign-off from qualified personnel. If any single test fails, the investigation and re-testing cycle begins again. What makes this even more challenging is that validation is not a one-time event. Every time a new sensor is added, software is updated, an alert threshold is changed, or a new cold room is brought online, parts of the validation process must be repeated.
For a growing pharmaceutical company, this becomes a near-constant operational burden, one that consumes significant time, delays new storage areas from being put into service, and requires specialist expertise that many organisations do not have in-house.
As a supplier of cold chain monitoring solutions, how does Lisaline Asia solve these challenges?
Since 1998, Lisaline Asia has been leading innovation in pharmaceutical cold chain monitoring. We have more than two decades of expertise in providing cold chain monitoring solutions. We are familiar with the challenges in the industry. To address these challenges, we partner with leading cold chain monitoring companies like Zebra Technologies, Dickson, TempMate, Degrite, and Hemokool. Our cold chain monitoring solutions are designed to solve the root problem rather than just address the temporary issue. Here’s how Lisaline solves the challenges:
Temperature controlled last mile delivery
Since most temperature excursions happen at the last step of the delivery, where they may be exposed to heat even for a few minutes. This issue is addressed with temperature-controlled shipping with Hemokool’s Cold Chain Boxes. Lisaline Asia supplies Hemokool’s Active Cooling Boxes and Passive Cooling Boxes. Depending on the time in transit and outside environmental conditions, you can choose between them. These portable refrigerators and freezers ensure temperature-controlled shipping till the products reach the end user.
Data Loggers with Battery Backup
Wireless data loggers such as Cobalt X from Dickson, supplied by Lisaline Asia, offer a battery backup option. In the event of a temporary power outage, the logger continues recording temperature and humidity using the backup battery. Tempmate’s GSM 4G data loggers and shipment data loggers with displays are fully battery-operated, so they remain unaffected by power interruptions. Similarly, Bluetooth data loggers such as Zebra ZS300 and Dickson Emerald are standalone battery-powered devices. Lisaline’s USB data logger Degrite S does not require a continuous electricity supply, ensuring that temporary power outages do not disrupt pharmaceutical cold chain monitoring.
Complete Supply Chain Cold Chain Monitoring
To solve the problem of multiple temperature monitoring systems making it difficult to track temperature excursions, Lisaline Asia supplies temperature indicators from Zebra Technologies. These temperature indicators monitor the temperature of products throughout the entire supply chain. They provide a clear visual signal of temperature exposure. They provide an irreversible signal when the temperature of the product goes beyond the safety parameters. The temperature indicator label is applied to the bottle and continues monitoring till the product’s shelf life.
The data loggers supplied by Lisaline Asia provide clear time-stamped data of the temperature readings. The data can be retrieved and downloaded into PDF/CSV reports. The time-stamped data allows you to pinpoint exactly where the excursion took place and identify patterns where breaches commonly occur. This helps you to make improvements and reroute shipment routes.
Automatic data loggers
As a modern upgrade to manual logging of temperature data, Lisaline Asia supplies automatic data loggers from brands like Dickson, Zebra, TempMate, and Degrite. These data loggers allow continuous monitoring of parameters like temperature, humidity and others at regular set intervals. The readings are recorded in the data logger’s internal memory.
In case of data loggers with cloud integration, the readings are sent to the cloud platform OCEAView for Dickson and Tempmate Cloud for Tempmate via different network systems like LoRaWAN, Bluetooth, 4G global connectivity, Wi-Fi, etc. You can view live time stamped readings on the cloud platform and download PDF/CSV reports, which provide you with clear proof whether the product is safe or not. In the case of USB data loggers, the time-stamped reports can be accessed just by plugging the data logger to a device once monitoring is done. These automatic systems do not require manual interference so there are no human errors.
High Accuracy and On-site Calibration
The data loggers supplied by Lisaline Asia are designed for high accuracy and show very little deviation from real environmental conditions. To ensure that the accuracy of sensors is maintained, Lisaline Asia offers on-site and in-lab calibration services for temperature, humidity, and CO2 sensors. It also provides both on-site and in-lab calibration in an ISO 17025/NABL-accredited facility. Our calibration process follows international standards and is carried out by a trained team to ensure accuracy, reliability, and compliance for cold chain monitoring applications.
Structured Validation Support
Lisaline Asia’s validation service removes the operational strain of proving compliance for pharmaceutical monitoring systems. Through structured IQ, OQ, and PQ support, Lisaline helps ensure that each system is properly installed, functions as required, and performs reliably in real-world conditions. This gives pharmaceutical companies the documented evidence they need while saving time, reducing rework, and making future changes easier to manage.
What is the role of IoT-based real-time alerts in pharmaceutical cold chain monitoring?
IoT-based real-time alerts play a crucial role in pharmaceutical cold chain monitoring by detecting temperature or humidity excursions as soon as they occur and enabling immediate corrective action. Since even a brief breach can affect product stability and potency, timely alerts help reduce spoilage, protect product quality, and minimise losses.
IoT-enabled cold chain temperature monitoring systems, such as wireless, GSM, or Bluetooth data loggers connected to cloud platforms like OCEAView or tempmate Cloud, continuously monitor environmental conditions and transmit readings at regular intervals. When measured values go outside the defined limits, the system automatically triggers alerts through SMS, email, app notifications, or other configured channels, allowing staff to respond quickly.
This real-time visibility improves shipment oversight, supports compliance documentation, and helps ensure that pharmaceutical products are delivered within safe temperature conditions.
Why choose Lisaline Asia for pharmaceutical cold chain monitoring?
Since 1998, Lisaline has been the pioneer of cold chain monitoring innovations in India. We transformed the industry by introducing the world’s first miniaturised temperature-exposure technology for vials as part of India’s Polio Eradication program. This landmark achievement gave us unmatched insight into the real-world challenges pharmaceutical companies face in cold chain monitoring — insight that continues to shape everything we do.
This deep expertise, combined with our access to globally trusted cold chain monitoring companies like Dickson, Tempmate, and Degrite, makes Lisaline Asia a leading choice for pharmaceutical cold chain monitoring solutions.
Lisaline Asia supplies a comprehensive range of cold chain monitoring products, including temperature indicators, wireless data loggers, USB data loggers, GSM data loggers, Bluetooth-enabled data loggers, shipment data loggers, and cold chain boxes. Each of these solutions is designed to maintain the integrity of your pharmaceutical products at every stage of the supply chain.
Lisaline Asia supports pharmaceutical cold chain monitoring through a wide range of technical services. These include temperature, humidity, and CO2 calibration, along with both on-site and in-lab calibration carried out in an ISO 17025/NABL-accredited facility. We also offer complete after-sales support, annual maintenance contracts, thermal mapping, validation services, device testing, and PQS/E006 testing for electronic and chemical temperature measuring devices — ensuring your cold chain operations remain compliant, accurate, and audit-ready.
Choose Lisaline for:
- 25+ years of cold chain expertise — a complete understanding of temperature-sensitive product protection built through decades of hands-on experience across India’s pharmaceutical and public health sectors.
- End-to-end cold chain monitoring solutions — covering every stage of your supply chain journey, from manufacturing to final delivery, so no link in your cold chain is left unmonitored.
- Access to globally trusted technologies — as an officially authorised supplier of leading international cold chain monitoring brands, we ensure reliability, regulatory compliance, and seamless integration at every touchpoint.
Conclusion
In conclusion, pharmaceutical cold chain monitoring is a critical operational, regulatory, and ethical requirement for every organisation that handles temperature-sensitive medicines, vaccines, and biologics. It involves continuous tracking and recording of environmental conditions across the entire supply chain, from manufacturing to final delivery. This is supported by real-time alerts, time-stamped data logging, and audit-ready compliance documentation. Without a reliable cold chain monitoring system in place, even a brief temperature excursion can compromise product potency, endanger patient safety, and result in significant financial and regulatory consequences.
Lisaline Asia supplies a complete range of pharmaceutical cold chain monitoring solutions, including temperature indicators, LoRaWAN wireless data loggers, USB data loggers, GSM 4G data loggers, Bluetooth data loggers, shipment data loggers, and cold chain boxes, along with technical services such as calibration, thermal mapping, validation, and PQS/E006 testing — all designed to maintain cold chain integrity at every stage of the supply chain.
To learn more about our pharmaceutical cold chain monitoring solutions or to discuss your requirements, contact our team today and visit our website for more details.